Marvin W Makinen
The University of Chicago, USA
Title: Inhibition of protein tyrosine phosphatase-1B in vitro and in vivo
Biography
Biography: Marvin W Makinen
Abstract
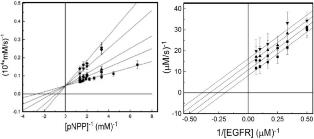
A large number of studies of protein tyrosine phospha-tases (PTPases) have been directed towards drug design for therapeutic intervention because of their critical roles in homeostasis and disorders of metabo-lism. In contrast to protein tyrosine kinases, virtually all inhibitors tested against PTPases exhibit only competi-tive behavior because of their consensus, active site sequence H/V-C-X 5-R-S/T, a condition leading to low specificity. Having identified protein tyrosine phospha-tase-1B (PTP1B ) as the target enzyme of the vanadyl (VO2+) chelate bis(acetylacetonato)oxidovanadium(IV) [VO(acac)2] in cultured 3T3-L1 adipocytes [Ou et al. (2005) J. Biol. Inorg. Chem. 10, 874-886], we have investigated the basis of inhibition by the VO2+-chelate through steady-state, kinetic investigations of the recom-binant human enzyme (residues 1– 321). Our results differ from investigations by others because we compared the influence of the chelate in the presence of the synthetic substrate p-nitrophenylphosphate (pNPP) and the phos-photyrosine-containing undecapeptide DADE-pYLIPQQG mimicking residues 988 – 998 of the epidermal growth factor receptor, a physiologically relevant substrate. We also compared the inhibitory behavior of VO(acac)2 to that of two other VO2+-chelates similarly known for their capacity to enhance cellular uptake of glucose as insulin mimetics. The results indicate that VO(acac)2 acts as a classical uncompetitive inhibitor in the presence of DADEpYLIPQQG but exhibits only apparent competitive inhibition with pNPP as substrate. Because uncompetitive inhibitors are more potent pharmacologically than compe-titive inhibitors, structural characterization of the site of uncompetitive binding of VO(acac)2 toPTP1B may provide a new approach to design of inhibitors of high specificity for therapeutic purposes.